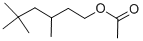
3-TRIDECANONE(CAS#1534-26-5)
1534-26-5 is 3-Tridecanone, an organic ketone compound widely used in fragrance, flavor, and scientific research.
Safety & Storage
- GHS Pictogram: GHS07 (Warning)
- Hazard Statements:
- H315: Causes skin irritation
- H319: Causes serious eye irritation
- H335: May cause respiratory irritation
- Personal Protection: It is recommended to wear protective goggles and gloves, and avoid inhalation of vapors or skin contact
- Storage Conditions: Store in a light-resistant, sealed container under inert atmosphere at room temperature to prevent oxidation
- WGK Germany: 3 (Severe hazard to aquatic environment)
Uses & Applications
- Fragrance & Flavor:
- Used in blending floral and fruity fragrance types, such as violet, cucumber, and watermelon notes, enhancing naturalness and complexity
- Due to its stable molecular structure, it can serve as a fragrance fixative
- Food Additive:
- Listed as FEMA GRAS (Generally Recognized as Safe), approved for use as a flavoring agent
- Recommended usage levels:
- Soft drinks, cold beverages: 0.15 mg/kg
- Candies, baked goods: 0.20 mg/kg
- Puddings, meat products, soups: 0.15 mg/kg
- Scientific Research & Biochemical Reagent:
- Serves as a biochemical reagent for olfactory receptor function studies and insect behavioral experiments (e.g., repellent activity research)
- Used as a standard or reference substance in life science research
- Industrial Raw Material:
- Used to synthesize ester derivatives, such as trans-2-nonenyl acetate, for further fragrance formulation
- Acts as an organic synthesis intermediate in constructing complex molecules
Synthesis Method
Prepared by reacting 1-bromo-2-nonen with acetic anhydride in acetic acid to form an acetate ester, followed by hydrolysis. Purification is achieved through vacuum distillation or column chromatography.
⚠️ Note: This product is a chemical substance, not for direct consumption or medicinal use. For industrial, research, or flavor formulation purposes only. Purchase requires appropriate qualifications.
Write your message here and send it to us