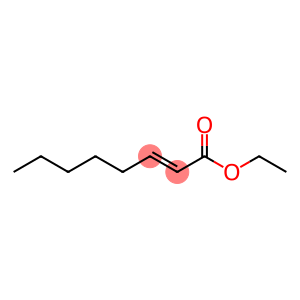
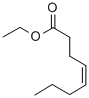
Ethyl Trans-2-Octenoate(CAS#7367-82-0)
Physical & Chemical Properties
- Appearance: Colorless to slightly yellow transparent liquid
- Odor Profile: Fruity, pear peel, tropical fruit, with a fatty nuance, classified as fruity-type (fruity)
- Density: 0.88–0.883 g/mL (25 °C)
- Boiling Point: 215.6 °C (760 mmHg) or 222 °C (literature value)
- Flash Point: 92.5 °C (open cup) or 209 °F (approx. 98.3 °C)
- Refractive Index: n₂₀/D 1.4410 (lit.)
- LogP: 2.686–3.97 (varies by source)
- Solubility: Soluble in ethanol, ether, chloroform, and other organic solvents
Safety & Storage
- GHS Pictogram: GHS05 (Corrosive)
- Hazard Statement:
- H314: Causes severe skin burns and eye damage
- Precautionary Statements:
- P260: Do not breathe vapors
- P280: Wear protective gloves, eye protection, and face protection
- P305 + P351 + P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing
- P310: Immediately call a poison center or doctor
- WGK Germany: 3 (Severe hazard to aquatic environment)
- Storage Conditions: Store at room temperature, in a dry, light-resistant sealed container, away from oxidizing agents and strong bases
Uses & Applications
- Fragrance & Flavor:
- Used as a fruity-type flavoring agent, especially in pear, tropical fruit, and green-type fragrance formulations
- Widely applied in beverages, candies, baked goods, and personal care products
- Serves as a precursor to natural aroma components, enhancing the authenticity and complexity of fragrances
- Food Additive:
- Listed as JECFA No. 1812, approved for use in food flavoring
- Not explicitly included in FEMA GRAS list, but recognized as a legal ingredient in flavor industry practice
- Research & Biochemical Reagent:
- Used in olfactory receptor studies and flavor metabolism research
- Acts as an organic synthesis intermediate for constructing more complex esters or functional molecules
- Industrial Raw Material:
- Used to synthesize other flavor derivatives, such as ethyl (E)-2-octenoate analogs or methyl esters, expanding its application in fragrance chemistry
Synthesis Method
Typically synthesized via acid-catalyzed esterification of trans-2-octenoic acid with ethanol. Alternative routes include olefin carbonylation or microbial fermentation. Purification is achieved through vacuum distillation or column chromatography.
⚠️ Note: This product is a chemical substance, not for direct consumption or medicinal use. For industrial, research, or flavor formulation purposes only. Purchase requires appropriate qualifications.
Write your message here and send it to us