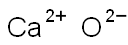Calcium oxide(CAS#73018-51-6)
Calcium oxide(CAS#73018-51-6) Introduction
Quicklime, also known as lime or burnt lime, is a common chemical substance with the chemical name calcium oxide (CaO). Below is an introduction to the properties, uses, production, and safety information of quicklime:
Properties:
- Appearance: White solid or white granules.
- Solubility: Quicklime reacts with water to produce calcium hydroxide (Ca(OH)₂), which has some solubility in water, forming an alkaline solution.
- Reactivity: Quicklime is strongly alkaline and reacts with acids to form salts and water.
Uses:
- Construction industry: Quicklime is widely used as a building material, for making lime mortar, calcining limestone, and preparing lime paste.
- Water treatment: Quicklime can be used to adjust the pH of acidic water, carrying out water treatment and purification.
- Agriculture: Quicklime is used to improve soil acidity and alkalinity, adjust soil pH, and promote plant growth.
- Metal smelting: Quicklime is also used in iron and steel production as well as in the smelting of non-metallic ores.
- Environmental protection: Quicklime can be used in sulfur and nitrogen removal from exhaust gases, reducing pollutant emissions.
Production:
In industrial production, quicklime is primarily prepared by calcining limestone at high temperatures. The limestone is first crushed and ground, then placed in a lime kiln for calcination. Under high temperatures, the calcium carbonate in limestone decomposes to produce calcium oxide.
Safety Information:
- Quicklime is alkaline and corrosive. In case of contact, rinse the affected area thoroughly with water immediately.
- When handling or storing quicklime, wear protective goggles, gloves, and respiratory protection to prevent direct contact or inhalation of dust.
- Avoid contact with organic or flammable materials to prevent possible reactions or fire.
- Avoid inhaling quicklime dust to prevent respiratory irritation or lung damage.
If needed, carefully read the relevant chemical safety data sheets.