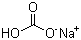Sodium bicarbonate(CAS#144-55-8)
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 1 |
| RTECS | VZ0950000 |
| TSCA | Yes |
| HS Code | 28363000 |
| Toxicity | LD50 orally in Rabbit: 4220 mg/kg |
Introduction
Soluble in water, almost insoluble in ethanol, lose carbon dioxide at about 50 ℃, and all heat becomes sodium carbonate at 100 ℃. It decomposes rapidly in weak acid, and its aqueous solution begins to decompose carbon dioxide and sodium carbonate at 20°C, and completely decomposes at the boiling point. Dissolved in 10 parts of water at 25 ℃ and 12 parts of water at about 18 ℃. The unstirred solution made of cold water 8.3 the pH value of 0.1mol/L aqueous solution freshly prepared with phenolphthalein test paper. Low toxicity, half lethal dose (rat, oral) 4420mg/kg.
Application
Sodium bicarbonate (CAS 144-55-8), commonly known as baking soda, is a versatile inorganic compound used across pharmaceutical, food, chemical, and environmental industries. As a pharmaceutical grade ingredient, it serves as an antacid for treating gastric acidity, an electrolyte replenisher in intravenous solutions, and an excipient in effervescent tablets. In food applications, it functions as a leavening agent in baked goods, a pH regulator, and a preservative. It is also widely used as a buffering agent in laboratory reagents, swimming pool water treatment, and as a cleaning and deodorizing agent. In industrial processes, sodium bicarbonate is applied in flue gas desulfurization (dry sorbent injection) to remove acidic gases, in leather tanning, textile dyeing, and as a fire extinguishing agent in Class C and K fire extinguishers. Its high purity grade ensures safety and performance in sensitive formulations. Xinchem offers custom synthesis, custom chemical synthesis, and contract manufacturing of high-purity sodium bicarbonate with flexible scaling from R&D to commercial tons. Contact us today for a competitive quote and reliable global supply.