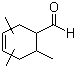
4-Chloro-5-iodo-7H-pyrrol[2,3-d]pyrimidine(CAS#123148-78-7)
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R22 - Harmful if swallowed R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37 - Wear suitable protective clothing and gloves. S37 - Wear suitable gloves. |
| WGK Germany | 3 |
| HS Code | 29335990 |
| Hazard Class | IRRITANT |
Introduction
4-Chloro-5-iodo-7H-pyrrolo [2,3-d] pyrimidine is an organic compound with the formula C8H3ClIN2. The following is an introduction to some of its properties, uses, methods and safety information:
Nature:
-4-Chloro-5-iodo-7H-pyrrolo [2,3-d] pyrimidine is a solid, usually colorless or pale yellow crystal.
-When dissolved in organic solvents such as methanol, chloroform and dichloromethane, it forms a solution that can be used in the laboratory.
Use:
-4-Chloro-5-iodo-7H-pyrrolo [2,3-d] pyrimidine is mainly used in scientific research and laboratory synthesis.
-As an organic raw material, it may also be used to synthesize other compounds or drugs.
Preparation Method:
-4-Chloro-5-iodo-7H-pyrrolo [2,3-d] pyrimidine can be synthesized by chemical reactions.
-A common preparation method is to react 2,3-diiodopyrimidine with 4-chloro-5-iodopyrrole in a solvent under alkaline conditions.
Safety Information:
-4-Chloro-5-iodo-7H-pyrrolo [2,3-d] pyrimidine has high safety under general conditions of use.
-However, when using and handling organic compounds, appropriate laboratory safety procedures should be followed.
-This compound may be irritating to the skin, eyes and respiratory system, so use caution when exposed and use appropriate personal protective equipment.
Please note that it is strongly recommended to use and handle organic compounds under professional guidance to ensure safety and correctness.
Application
4-Chloro-5-iodo-7H-pyrrolo[2,3-d]pyrimidine (CAS 123148-78-7) is a highly functionalized heterocyclic building block bearing two orthogonal halogen leaving groups (chlorine at position 4, iodine at position 5) on the pyrrolo[2,3-d]pyrimidine core. This unique dihalogenated scaffold enables sequential, site-selective cross-coupling reactions, allowing the introduction of diverse aryl, heteroaryl, or alkyl substituents via Suzuki-Miyaura, Buchwald-Hartwig, Sonogashira, or Ullmann couplings. The chlorine at C4 is more reactive toward nucleophilic aromatic substitution, while the iodine at C5 is preferentially engaged in palladium-catalyzed cross-couplings. As a purine bioisostere, this compound is extensively used in medicinal chemistry for the synthesis of highly selective kinase inhibitors targeting cancer, inflammation, and autoimmune diseases (e.g., JAK, BTK, CDK, and TBK1). Its orthogonal reactivity pattern enables the construction of complex, drug-like libraries and facilitates rapid structure-activity relationship (SAR) studies. High purity ensures reproducible results in high-throughput screening and scalable pharmaceutical synthesis. Xinchem offers custom synthesis, custom chemical synthesis, and contract manufacturing of this key intermediate with flexible scaling from R&D to commercial tons. Contact us today for a competitive quote and reliable global supply.


![4-Chloro-5-iodo-7H-pyrrol[2,3-d]pyrimidine(CAS#123148-78-7) Featured Image](https://www.xinchem.com/uploads/4-Chloro-5-iodo-7H-pyrrol23-dpyrimidine.png)