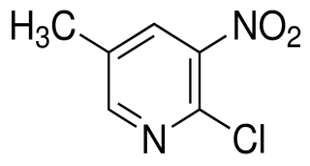
6-Iodo-1H-Pyrrolo[3,2-B]Pyridine(CAS#1015609-75-2)
Introduction
6-Iodo-1H-pyrrolo[3,2-b]pyridine is an organic compound. The following is an introduction to the properties, uses, preparation methods and safety information of the compound:Quality:6-Iodo-1H-pyrrolo[3,2-b]pyridine is a solid that can form crystals. It is a brown to dark red crystalline solid with strong hygroscopicity of ammonia and dimethyl sulfoxide. The compound is stable at room temperature but easily decomposes at high temperatures.Use:6-Iodo-1H-pyrrolo[3,2-b]pyridine has certain application value in the field of chemical industry. It is often used as an intermediate in organic synthesis for the synthesis of other organic compounds. It can also be used as an organic electronic material and a coordination chemistry system.Method:The method for preparing 6-iodo-1H-pyrrolo[3,2-b]pyridine can be one of the following:Under suitable reaction conditions, 1H-pyrrolo[3,2-b]pyridine is reacted with iodine atoms.Pyrrolo[3,2-b]pyridine is introduced in the N-iodation reaction.Safety Information:The compound can be irritating to the eyes and skin, and appropriate personal protective equipment such as protective glasses, lab gloves, and lab coats must be worn when handling.Avoid inhaling or ingesting the compound, and if inhaled or ingested, remove it immediately from fresh air and seek medical attention if discomfort occurs.In order to avoid environmental pollution and health risks, the compound and its waste need to be properly disposed of, in accordance with local regulations.
Application
6-Iodo-1H-pyrrolo[3,2-b]pyridine (CAS 1015609-75-2) is a halogenated azaindole derivative featuring an iodine atom at the 6-position of the fused pyrrole-pyridine core. As a purine bioisostere, this heterocyclic scaffold is widely employed in medicinal chemistry and drug discovery for the synthesis of highly selective kinase inhibitors targeting cancer, inflammatory diseases, and autoimmune disorders (e.g., JAK, BTK, CDK, TBK1, and PI3K inhibitors). The iodine atom serves as an excellent handle for palladium-catalyzed cross-coupling reactions, including Suzuki-Miyaura, Buchwald-Hartwig, Sonogashira, and Ullmann couplings, enabling rapid introduction of diverse aryl, heteroaryl, or alkyl substituents for structure-activity relationship (SAR) studies. The NH group allows further alkylation or acylation to fine-tune physicochemical properties and target selectivity. This building block is also utilized in the synthesis of antiviral agents, CNS drugs, and agrochemicals, as well as for the construction of focused compound libraries in high-throughput screening. Its high purity (≥98%) ensures reproducible results in complex synthetic routes and scalable pharmaceutical processes. Xinchem offers custom synthesis, custom chemical synthesis, and contract manufacturing of 6-iodo-1H-pyrrolo[3,2-b]pyridine with flexible scaling from R&D to commercial tons. Contact us today for a competitive quote and reliable global supply.


![6-Iodo-1H-Pyrrolo[3,2-B]Pyridine(CAS#1015609-75-2) Featured Image](https://www.xinchem.com/uploads/6-Iodo-1H-Pyrrolo32-BPyridine.png)