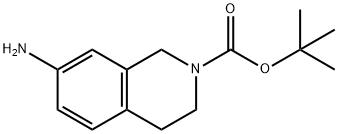
7-AMINO-3,4-DIHYDRO-1H-ISOQUINOLINE-2-CARBOXYLIC ACID TERT-BUTYL ESTER(CAS#171049-41-5)
| Risk Codes | R22 - Harmful if swallowed R50 - Very Toxic to aquatic organisms |
| Safety Description | 61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN2811 |
Introduction
T-butyl-7-amino-3,4-dihydroisoquinoline-2(1H)-formic acid is an organic compound. The following is an overview of the compound's properties and uses, preparation methods, and safety information:Quality:- Appearance: Usually colorless or light yellow solid.- Solubility: Soluble in ethanol, dimethyl sulfoxide and chloroform, slightly soluble in water.- pH: Alkaline.Use:Method:- The compound can be synthesized by different synthetic routes, one of the commonly used methods is the esterification of 7-amino-3,4-dihydroisoquinoline-2(1H)-formic acid with tert-butyl protecting groups.Safety Information:- The specific safety information of tert-butyl-7-amino-3,4-dihydroisoquinoline-2(1H)-formic acid may depend on the purity and form of the compound, and the safety data sheet (SDS) of the chemical should be read carefully and followed before use.- If not handled or disposed of properly, the compound may pose a hazard to the environment and should be handled and disposed of in accordance with local regulations.
Application
7-Amino-3,4-dihydro-1H-isoquinoline-2-carboxylic acid tert-butyl ester (CAS 171049-41-5) is a Boc-protected tetrahydroisoquinoline (THIQ) derivative featuring a free primary amine at the 7-position. This versatile building block is widely used in medicinal chemistry and pharmaceutical research for the synthesis of central nervous system (CNS) agents, including potential treatments for Parkinson’s disease, Alzheimer’s disease, depression, and anxiety disorders. The Boc group enables selective deprotection under mild acidic conditions, while the primary amine allows amide, urea, or sulfonamide formation, facilitating rapid structure-activity relationship (SAR) studies. This intermediate also serves as a key precursor for kinase inhibitors targeting cancer, inflammation, and neurological disorders, as well as for GPCR modulators and neuroprotective agents. Its rigid, saturated THIQ core improves metabolic stability and blood-brain barrier penetration. High purity ensures reproducible results in high-throughput screening and scalable GMP synthesis. Xinchem offers custom synthesis, custom chemical synthesis, and contract manufacturing of this compound with flexible scaling from R&D to commercial tons. Contact us today for a competitive quote and reliable global supply.


![4-chloro-1H-pyrazolo[4 3-c]pyridine(CAS# 871836-51-0)](https://www.xinchem.com/uploads/4chloro1Hpyrazolo43cpyridine.png)